|
| 7. Protons, Neutrons, Electrons |
|
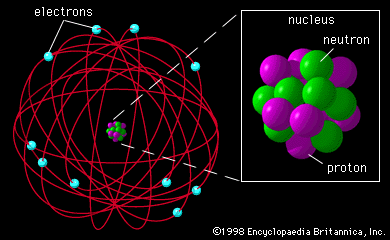 |
In the twentieth century an atomic model has been developed that can describe the complexity of the many different elements.
An atom consists in this model of a nucleus containing protons and neutrons, around which electrons are orbiting. Protons have a positive charge, electrons an equal, but negative charge, while neutrons are uncharged. Protons and neutrons (called nucleons) have about the same mass, the mass of an electron is almost negligeable. The electromagnetic force keeps the electrons in their orbit, as the gravitational force keep the planets in their orbit around the sun. Another force, the Strong Nuclear Force, exists between nucleons and keeps them together, against the strong repulsive electromagnetic force between the protons.
|
The field of atomic and nuclear physics is a complicated one. Quantum Mechanics is needed, even Relativity Theory. On the microscopic scale of atoms, particles behave like waves and waves can act as particles. A visualisation of an atom as given in the picture above, is misleading and very inaccurate. An atom is NOT a miniature solar system. Fortunately, for our story, we do not need too many details. Characteristic for an element is its number of protons in the nucleus. Let us have a look at the elements which are important for the human body. The most simple atom is Hydrogen, with a single proton it its nucleus and one electron in orbit
The next simple one is Helium, which is NOT present in our body and even rare on Earth, although the second most common in the Universe! It consists of two protons and two neutrons in its nucleus, with two orbiting electrons. Actually there exists another Helium, with two protons, but only one single neutron! Atoms with the same number of protons and a different number of neutrons, are called isotopes. In the next paragraph we will see that isotopes are not always stable, they can be radioactive.
|
A few examples. Let us forget about the electrons for the time being and concentrate on the atomic nucleus.
Carbon has 6 protons in its nucleus, Oxygen 8 protons and Calcium, the major constituent of our bones and teeth, has 20 protons.
Carbon has two stable isotopes, with 6 and 7 neutrons, resp. We write C-12 and C-13. Oxygen has three stable istopes, O-16, O-17 and O-18. And Calcium has even four, Ca-40, Ca-42, C43 and Ca-44 . Can we go on with more and more protons?
Below the so-called nuclides map is shown. The horizontal axis gives the number of neutrons, the vertical axis the number of protons. The map has been cut into three pieces. Stable isotopes are black, the radioactive ones are colored. When you double-click on the map, you can see all its details, but be prepared for a long download time, the file is 5 MB!
|
 | |
|
So it is just a matter of numbers. Does the nucleus contain 26 protons? Then it is an iron isotope. 82 protons? Lead. Suppose it was possible to remove 3 protons from a lead nucleus. Then you would get gold (atomic number 79)! The dream of the alchemist reduced to numbers.
How far can we go? Not very far.
When the number of protons becomes too large, the element becomes instable, radioactive. In the next chapter we will see why. Uranium, with 92 protons, has the highest atomic number of the naturally occurring elements.
But is not completely stable. The half-life of Uranium-280 is about 4.5 billion years. |  |
|
|